BSC Class IV APIs in oral formulations
APIs of BSC Class IV in oral formulations represent one of four groups following the Biopharmaceutical Classification System (BCS). The BCS predicts potential effects of oral formulations on the human drug oral bioavailability. Thereby, it is a scientific tool differentiating the drug substances on the basis of solubility and intestinal permeability under prescribed condition. While the solubility classification is based on a United States Pharmacopoeia (USP) aperture, the classification of permeability is based on a comparison to the intravenous injection.
| Permeability | Class I
High solubility High permeability |
Class II
Low solubility High permeability |
| Class III
High solubility Low permeability |
Class IV
Low solubility Low permeability |
|
| Solubility | ||
Table 1: Biopharmaceutics Classification System (BCS).
BSC Class IV
BSC Class IV drugs are characterized by a low solubility and a low permeability (Table 1). Typical drugs are Verapamil HCL, Propiverine HCL, Papaverine, or Dipyridamole. These APIs are the most challenging for product and process development. The low solubility above approximately pH 4.5 is turning into a fairly good solubility in the acidic range.
Due to low water solubility in the neutral pH range of BSC class IV drugs, a potential substrate in pharmaceutical formulations for oral drugs needs to change the pH into acidic regimes so that solubility of API is positively affected. In case of MUPS (Multiple Unit Pellet Systems) or capsule dosage forms, pellets of Tartaric Acid pellets – short: TAP – are the first choice. Depending on the pellet size, they serve as starter beads in MUPS formulations (smaller than about 800 µm) or in capsules (larger than approx. 800 µm). Insider-tip: have an sealed coating as isolation layer between the TAP starter pellet and the API layer, to prevent incompatibility issues with the API and stability issues with the product.

Figure 1: Pellet sizes for potential BSC Class IV MUPS and capsule formulations.
Tartaric acid spheres or pellets for MUPS formulations:
TAP 850-1000 (850-1000 µm)
TAP 700 (600-800 µm)
Tartaric acid spheres or pellets for BSC Class IV capsule formulations:
TAP 600 (500-700 µm)
TAP 500 (400-600 µm)
TAP 400 (300-500 µm)
TAP 200 (100-350 µm)
Further information
Are you working with BSC Class I actives? Read more here.

 ingredientpharm
ingredientpharm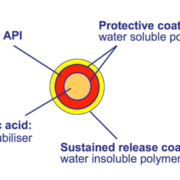
 ingredientpharm
ingredientpharm
 ingredientpharm
ingredientpharm